Transcranial Magnetic Stimulation (TMS) has been researched for over 30 years. In 2008, it received FDA approval in the U.S. for the treatment of major depressive disorder in adults who did not find relief from medications. Given its proven effectiveness, researchers continue investigating the benefits of TMS for various psychiatric and neurological conditions. With that in mind, let’s explore the three TMS protocols we offer at Scenic City Neurotherapy.
Repetitive Transcranial Magnetic Stimulation (rTMS) is a series of repetitive, brief magnetic pulses that stimulate targeted neurons in the brain. These magnetic pulses induce an electrical current in the brain, which stimulates the targeted nerve cells, thus optimizing communication and restoring brain activity to normal levels with little to no side effects.
Intermittent Theta Burst Stimulation (iTBS), or the SNT Protocol, is an evolution of TMS therapy that uses more frequent, lower intensity, stimulation 10 times/day for 5 consecutive days and has shown to produce a significantly higher rate of patient-reported remission of symptoms than Classic rTMS protocols.
At Scenic City Neurotherapy, both Classic rTMS and iTBS (SNT protocol) have three ketamine boosters built in to the treatment regimen to enhance global optimization and produce a more comprehensive, long lasting remission of symptoms.
ONE-D is a one-day TMS protocol for depression, achieving over 90% response rates. Developed by AMPA, the protocol begins with plasticity priming, followed by 20 iTBS sessions over 9.5 hours, one every 30 minutes. By combining targeted brain stimulation with plasticity priming, the ONE-D protocol is transforming depression care, making it faster, more effective, and more accessible than ever before.
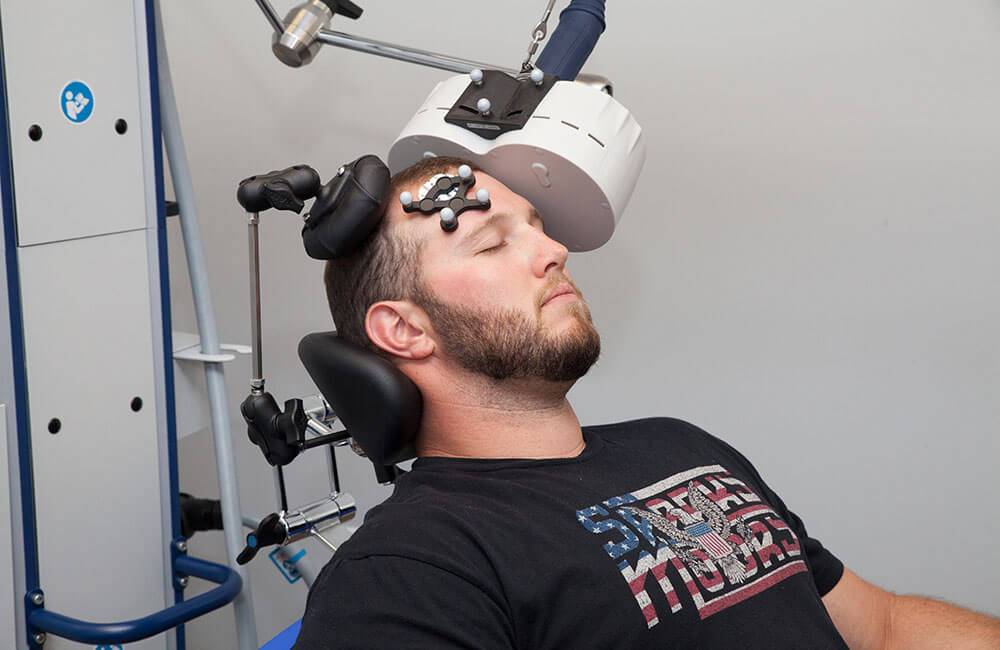
Patient outcomes are our number one priority. This is why we chose the most advanced FDA-approved rTMS therapy system that offers neuronavigation. Neuronavigation helps to ensure precise coil positioning with every treatment – thus enhancing outcomes for a better quality of life.
The Magstim name is at the forefront of Repetitive Transcranial Magnetic Stimulation (rTMS) from its conception as a widely used research tool to its revolution for treatment in the clinic setting. At Scenic City Neurotherapy, treatment doesn’t require the use of head caps.
At Scenic City Neurotherapy, our goal is to offer the possibility of remission to all patients struggling with treatment-resistant depression. We understand that attending the daily treatment sessions for six weeks, as required by rTMS, is just not possible for some individuals. Factors such as work obligations, geographic distance, or other obstacles could make it difficult to commit to this schedule.
To address this, Stanford has developed a protocol featuring intermittent theta burst stimulation (iTBS). It allows patients to receive treatment in as little as 10 minutes for a much shorter period – five days – instead of six to nine weeks. The SNT Protocol has a 90%+ rate of remission of depressive symptoms. While the SNT protocol requires multiple treatments in a day, the greatest benefit to Stanford’s protocol is the ability to achieve remission in as little as five days.
The SNT Protocol is not covered by insurance yet. However, at Scenic City Neurotherapy, the SNT Protocol is one of the most affordable TMS treatment options in the U.S. Why? Because our mission is to ensure access to novel treatments for people suffering from depression and to facilitate a better quality of life.
Learn more bout our Ketamine-Assisted TMS Protocols on our blog.
Repetitive transcranial magnetic stimulation (rTMS therapy) is a simple, non-invasive, non-drug, therapy that does not require downtime. Patients can immediately return to their normal activities of daily living.
A standard course of treatment consists of a total of 36 treatment sessions over a course of six weeks, with the last six rTMS treatment sessions titrated over a three-week period.
It is important to understand that rTMS is not a permanent cure for depression. However, rTMS does offer long-term symptom relief and remission for nearly 70% of patients. Maintenance treatments are recommended in order to maintain optimal benefits.
There are nearly 17 million adults in the United States alone that have experienced at least one major depressive episode in the past year. With the outbreak of COVID-19, the incidence of depression and anxiety has escalated. Although awareness is improving, it still has a long way to go.
Depression, also known as major depressive disorder or clinical depression (and often referred to as treatment resistant depression), is more than just a feeling of sadness. In fact, depression is a serious illness with multiple symptoms that can have a significant impact on both your mental and physical health.
Published by the American Psychiatric Association, the DSM-5 explains that a diagnosis of depression requires at least five (5) or more of the below symptoms in the same 2-week period. Symptoms include:
Sadly, medications do not always help, and often cause other debilitating symptoms due to debilitating side effects. Long-term use of psychiatric medications can also cause undesired, permanent side effects, as well as horrible withdrawal symptoms when trying to titrate off of them.
When medications have been ineffective, or for those who do not wish to take medications, TMS can be a safe and effective alternative that offers long-term relief.
There are many advantages to TMS therapy as a treatment option for depression without any other significant diagnoses. A few advantages of TMS with StimGuide include:
TMS bypasses common side effects of antidepressants such as:
TMS avoids the unpleasant side effects of ECT therapy such as:
You can achieve long term relief in as little as five days without the shock or induction of a seizure like with ECT.
TMS has been researched for over 30 years. In 2008, it received FDA approval in the U.S. for the treatment of major depressive disorder in adults who did not find relief from medications. Given its proven effectiveness, researchers continue investigating the benefits of TMS for various psychiatric and neurological conditions. At Scenic City Neurotherapy, we offer the rTMS, iTBS, and ONE-D protocols, as well as ketamine-assisted TMS protocols.
Those experiencing treatment-resistant depression who have not improved with medication often benefit from TMS.
TMS targets the prefrontal cortex, sending pulses that activate underperforming networks responsible for motivation and emotional balance. Over time, this rewires the brain toward healthier patterns.
Most people feel a tapping sensation on the scalp during treatment and have no downtime afterward. Side effects are typically mild and may include brief scalp sensitivity or a light headache.
Yes. TMS is a non-medication, evidence-based treatment that uses gentle magnetic stimulation to improve mood, focus, and overall brain health without systemic side effects.
At Scenic City Neurotherapy, patients report 90% or greater remission of their depressive symptoms when completing TMS using either the SNT or the ONE-D treatment approaches.
At Scenic City Neurotherapy in Chattanooga, TMS is provided in a controlled, evidence-based setting designed to help patients improve long-term brain health and reduce depressive symptoms.
Transcranial Magnetic Stimulation (TMS) is insurance-reimbursable, and we help patients navigate that process. Insurance does not typically cover Minimally Stimulated Ketamine Infusion Therapy (MSKIT®), though many patients use health savings accounts, and we offer flexible payment options through CareCredit. Scenic City Neurotherapy also provides the SCN Service Program, which offers scholarships for those who qualify. Additionally, we are in network with Veteran Affairs, and veterans only need a VA referral to begin care. Our team will walk you through every option available and provide any documentation needed for reimbursement attempts.